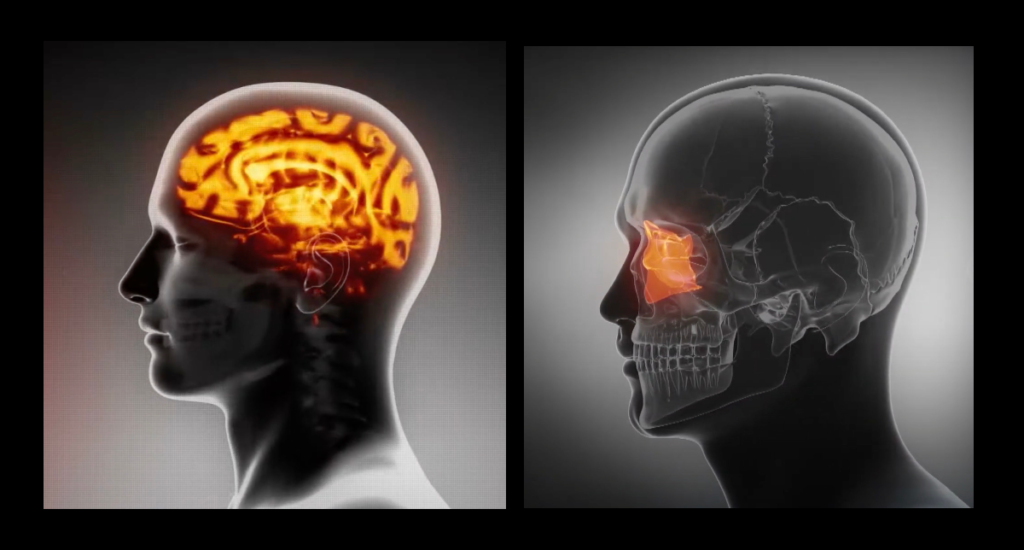
Leucadia Therapeutics has developed AI algorithms that allow neuroscientists to analyze enormous datasets. This new approach incorporates cutting-edge deep learning applied to an advanced method for ultra-resolution contrast-enhanced micro-CT, a novel animal model, and a large clinical study designed to produce highly curated data for machine learning.
For the past 25 years, Alzheimer’s disease researchers have viewed plaques and tangles pathology as the cause of Alzheimer’s, which has led to an unbroken string of failed clinical trials. In 2014, neuroscientist Doug Ethell, PhD, published a new hypothesis about the trigger for Alzheimer’s and related dementias. In 2015, he founded Leucadia Therapeutics to develop a therapy based on his hypothesis. Leucadia’s research has shown that plaques and tangles are effects of a more serious underlying condition that triggers the formation of those pathological features.
On October 5, 2021, Dr. Ethell’s team reported on three studies that confirm his hypothesis. A manuscript entitled Impairment of CSF Egress through the Cribriform Plate plays an Apical Role in Alzheimer’s disease Etiology is in peer-review and is available on MedRXiv’s preprint server. This research has implications for diagnosing, treating, and preventing Alzheimer’s and provides conclusive evidence that cribriform plate aging plays a critical role in Alzheimer’s disease etiology. These findings open the door to new opportunities for prediction and treatment years before dementia and Alzheimer’s become irreversible.
How the Brain is Affected by Cribriform Plate Ossification
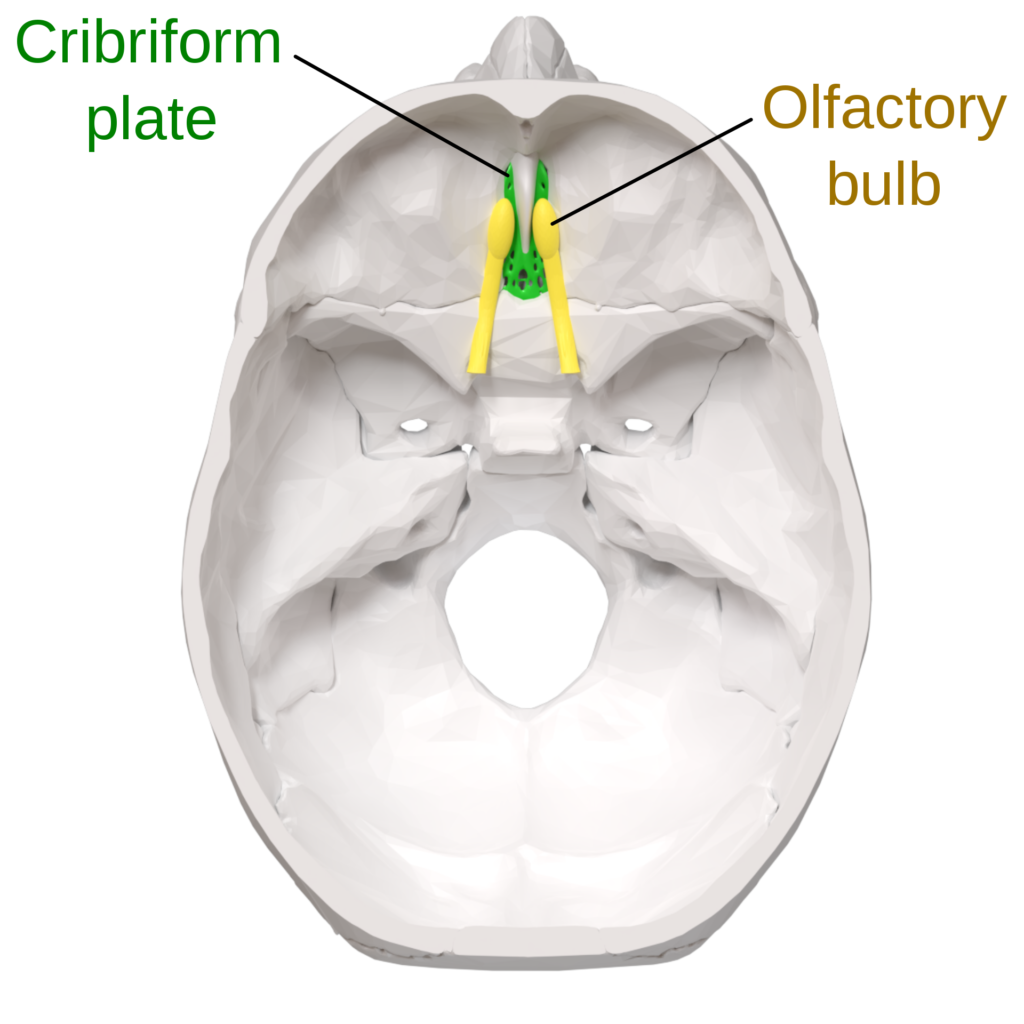
The cribriform plate is a porous bony structure located in the roof of the nasal cavity. The plate contains two deep pockets called fossa and many holes called olfactory foramina. Olfactory nerves that transmit the sense of smell pass through these holes. The cribriform plate is an outflow route the brain uses to clear out waste in cerebrospinal fluid (CSF). About half a liter of CSF is produced by the brain each day, but only about 1/20 of it drains through the cribriform plate. However, that small amount of CSF is responsible for clearing brain regions that are critical for making new memories and orienting us in the world. As humans age, the cribriform plate becomes ossified and less porous. The small holes close up and restrict the flow of cerebrospinal fluid. As less and less of this fluid is drained out of the brain, waste and toxins accumulate in the upstream brain regions responsible for memory. These waste-products form a slough (an area of dead tissue) where plaques form and neurons form tangles—two key hallmarks of Alzheimer’s disease. Ethell’s research indicates that Alzheimer’s disease pathology result from reductions in cerebrospinal fluid drainage across the cribriform plate.
“It’s one thing to tell someone that they will develop dementia or Alzheimer’s in 5 or 10 years, and something else to offer them a solution.”
Doug Ethell, PhD, Founder and CEO, Leucadia Therapeutics
CSF Intervention To Reverse Early Alzheimer’s
Cerebrospinal fluid (CSF) clears toxic metabolites from intercellular spaces in the brain, similar to the way that the lymphatic system clears toxins and other impurities from the rest of the body. As the first regions of the brain impacted by Alzheimer’s disease are cleared by CSF that drains across the cribriform plate, things that affect that structure may increase dementia risk. Aging and life events that accelerate cribriform plate occlusion and reduce CSF-mediated clearance from those brain regions, increase Alzheimer’s disease risk, including the formation of plaques and tangles. Leucadia Therapeutics is preparing for a clinical trial in 2022 that will use an implantable device to restore CSF drainage across the cribriform plate.

With increasing life-expectancy, Dr. Ethell believes his device will become as common as a pace-maker. This well-founded approach should reverse early mild cognitive impairment and prevent Alzheimer’s disease from occurring at all. Leucadia’s patented Arethusta® technology restores CSF flow across the cribriform plate, improving the clearance of toxic metabolites from the earliest regions of the brain to be affected by Alzheimer’s disease. The name Arethusa is based on a water nymph from Greek mythology who Artemis saved by turning her into a hidden underground stream. Leucadia’s device will create an under-the-tissue channel to drain fluid from the brain. The Arethusta device will be introduced through the nose as the cribriform plate is at the top of the nasal cavity. In clinical studies in ferrets, researchers artificially blocked drainage through the plate. That blockage induced cognitive impairment and atrophy in the same brain regions where Alzheimer’s disease strikes first. CSF management using shunts for hydrocephalus is performed by neurosurgeons everyday, but Arethusta takes CSF from a very different place for a much more specific effect.
Using Deep Learning To Analyze Changes In Cribriform Plate Morphology
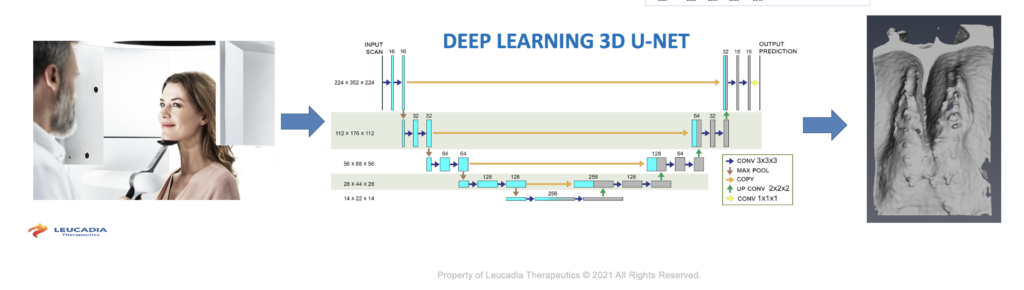
Leucadia Therapeutics has developed AI algorithms that allow researchers to analyze enormous datasets. This new approach incorporates cutting-edge deep learning applied to an advanced method for ultra-resolution contrast-enhanced micro-CT, a novel animal model, and a large clinical study designed to produce highly curated data for machine learning. Scientists at Leucadia combined these methods to compare changes in cribriform plate morphology in Alzheimer’s disease.

3D U-Net segmentation is an architecture based on the Convolutional Neural Network, which is typically used to classify labels. However, in medical imaging 3D U-Net can do more than just classify. It contains localization to predict the class label of each pixel by providing a local region around that pixel as an input. Machine learning algorithms treat voxels as normalized floating-point calculations, so combinations of voxels that denote edges and boundaries in a 65,000 gray level scan can be normalized to predict bony structures in a cone-beam CT with lower resolution.

Dr. Ethell and his team used 3D-UNet to train models with extremely high-resolution samples from post-mortem micro-CT samples. He then used those models to predict bone patterns from low-dose clinical CT scans that have lower resolution. The image on the left is a 20 second cone-beam CT scan image. Note how the bone detail is hard to see. The information is there, but it is spread across thousands of gray levels. The image on the right is how the trained AI model predicted bone from the scan on the left. Note the detail including apertures. CT scans produce 3,000 to 65,000 gray levels but the human eye can distinguish fewer than 20 gradations and manual segmentation of large 3D volumes is difficult and time-consuming. AI algorithms developed by Leucadia identify edges, boundaries, and holes to reveal where apertures occur and how large they are using a low-dose cone-beam CT scan.
Next Steps
This work lays the foundation for Leucadia’s application to the FDA for a clinical trial of Arethusta set to begin in 2022. Current investors include David Gobel, Jim Mellon, Methuselah Fund, and Methuselah Foundation. The company is planning a Series A raise in 2022 to fund the Arethusta clinical trial.
Margaretta Colangelo serves on the advisory board of Leucadia Therapeutics.
